Believing in the power of research
At Ferring we believe in the power of research and the need to go where the research takes us. We are passionate and curious about science.
Ferring invests heavily in the process of discovering and developing new medicines through its research and development centers and through research collaboration with scientific institutes, as well as biotechnology and pharmaceutical companies throughout the world.
Ferring’s R&D activities are focused on the development of medicines within our key therapeutic areas: Reproductive Medicine & Maternal health, Urology & Uro-oncology, Gastroenterology & Immunology. Our focus is on discovering differentiated therapies that address unmet needs in our key therapeutic areas through deep understanding of disease biology and the application of enabling novel technologies.
Ferring has R&D centres in 10 countries (China, Denmark, India, Israel, Japan, Scotland, one in Switzerland and three in the US).
The main pharmaceutical development hubs are in Copenhagen, Denmark and San Diego, US. These sites drive and execute innovation and development of new molecular entities as well as further development of our existing in-line products to better meet patient needs and address new disease areas.
Our scientific approach
At Ferring, we believe in the power of research and we are committed to providing innovative solutions to help people live better lives. Our researchers focus on striving to best address the needs of patients.
Our R&D activities are focused on the discovery and development of transformational medicines in our key therapeutic areas regardless of their source. Every day our scientists are charting new territory and continuing our commitment to innovation that has created exceptional medicines that improved the lives of patients.
Microbiome
Ferring has a keen interest in the microbiome and is seeking treatment solutions across our key therapeutic areas in the microbiome space.
Learn more about our work in the microbiome.

Recombinant protein and mAbs
Ferring is specialised in researching and developing biotechnology derived medicines.

Small molecules
Ferring aims to develop innovative medicines based on small molecules together with partners.
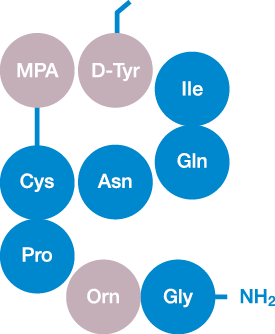
Building on our scientific heritage within peptide technology
Ferring has extensive knowledge on peptide-based drugs, these are at the heart of Ferring’s R&D activities and the history of the company.
Ferring’s peptide technology and know-how combines naturally-occurring, DNA-coded, amino acids with large collections of proprietary synthetic amino acids to “improve on nature” by overcoming the typical limitations of endogenous peptides and hormones, primarily pleiotropic and short-lived actions.
The resulting “drug-like” molecules retain the key advantages of naturally-occurring peptides and hormones (high potency, high efficacy and high safety), while offering improved pharmacodynamics (receptor and function selectivity), pharmacokinetics (enzymatic stability, clearance, half-life and duration of action) and pharmaceutical properties (solubility and stability).
Recombinant protein and biologics
Ferring is also specialised in researching and developing biotechnology-derived medicines, such as recombinant proteins and other biologics. In addition, we are investing in building capabilities for monoclonal antibodies.
Innovative science
We rely on the knowledge and experience of our talented scientists, who use their passion and creativity to generate innovative drugs for patients.
Our scientific approach doesn’t stand still, and we are embedding data science and digital technologies across Ferring to transform the way we innovate in R&D. We are applying computational biology, artificial intelligence and machine learning within our drug discovery approach.
We are continuously investing in new technologies to develop a new era of science that reflects our innovative mindset.
We are building upon our heritage and our extensive knowledge in recombinant protein technologies to develop transformative medicine.
Drug delivery
Drug delivery is how the treatment is administered to the patient. Drug delivery principles are traditionally described as parenteral and oral.
The preferred delivery principle for patients is the oral route. However, the nature of the drug often makes it impossible so that a parenteral formulation principle has to be chosen. Parenteral delivery systems can be classified in different ways but are typically divided into sterile (e.g. injectables) and non-sterile products (e.g. rectal, vaginal, transdermal or nasal formulations).
Ferring’s fundamental principle is to develop drug delivery systems offering the most patient-friendly dosage form possible with regards to ease of administration and convenience.
Discover our areas of research


